A new direct approach to pancreatic cancer therapy
and other solid tumours
Expert perspectives
Leading physicians share their clinical insights on YntraDose
Innovation
The YntraDose Solution
Technology
How YntraDose works
Tailored for use by Nuclear Physicians and Interventional Radiologists, YntraDose allow to provide patients with a personalised, targeted and precise radiotherapy treatment that can be achieved on a tumour-by-tumour and patient-by-patient basis.
Localize
Locate lesion or tumour injection site.
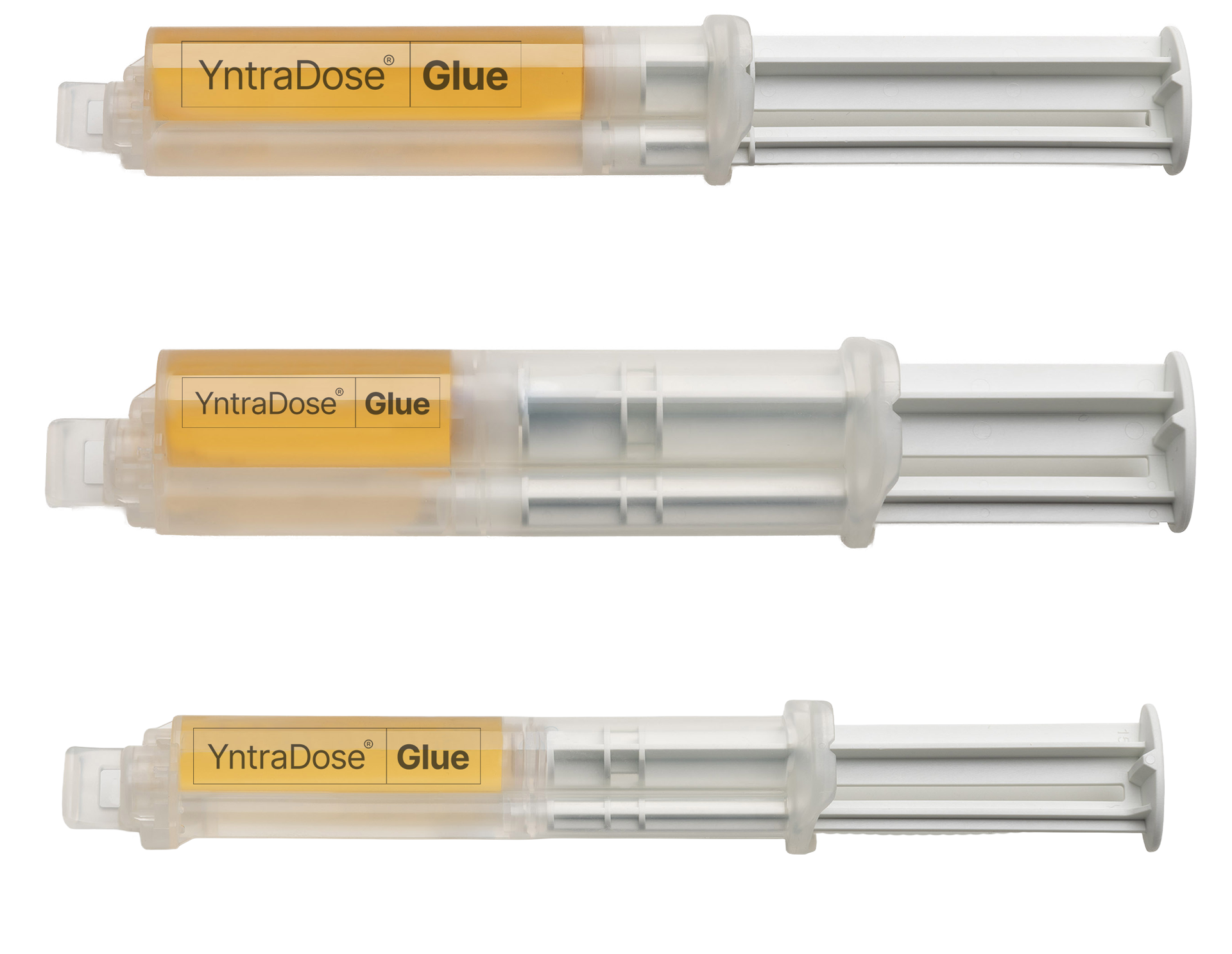
Prepare
YntraDose-90 added to the YntraDose-Glue syringe.
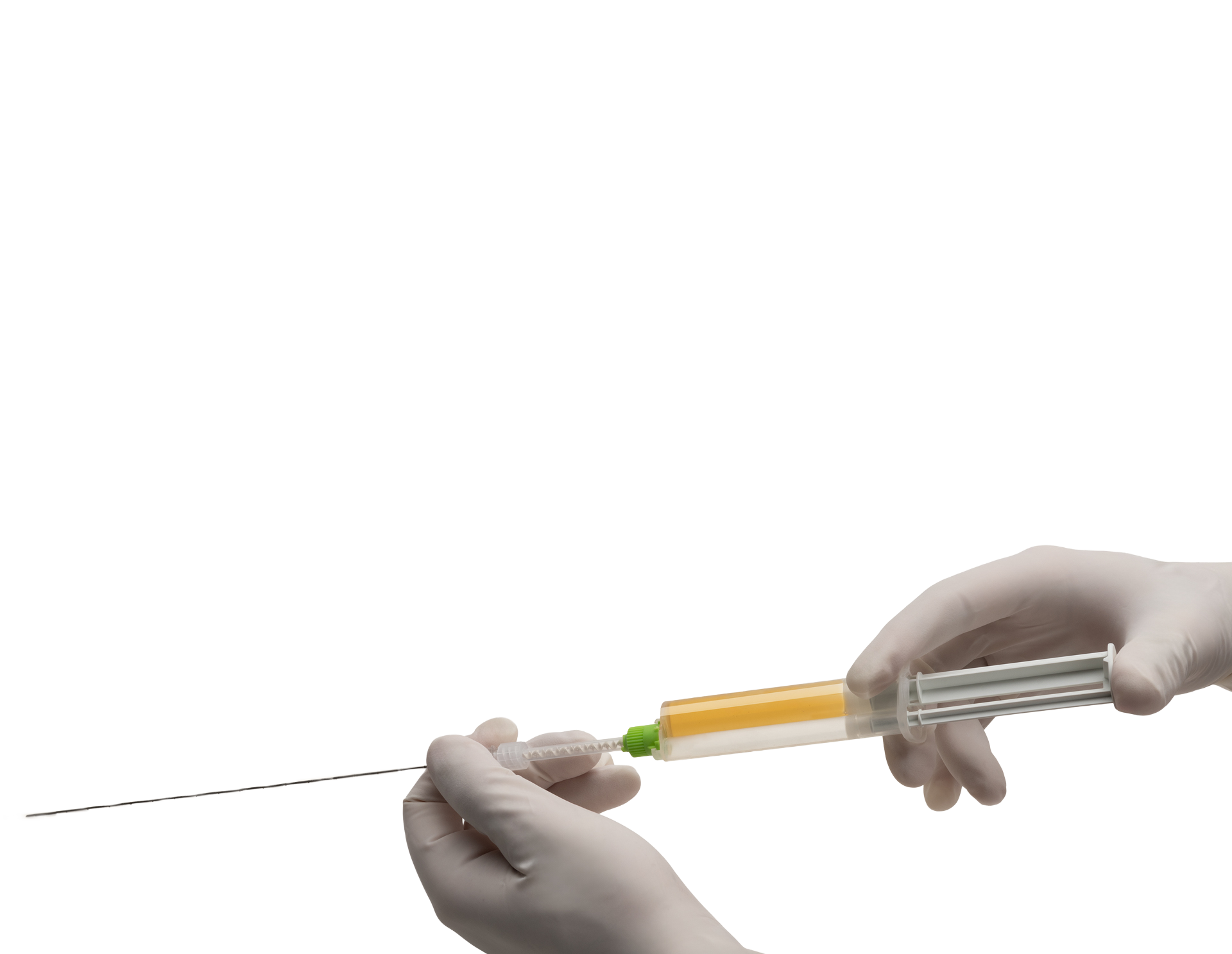
Treat
Attached YntraDose-Deliver injection system to YntraDose Syringe.
Assess the outcome
Assess focalized signal of implantation of YntraDose into injection site.
Pre-Clinical & Clinical
Publications
- Modelling a new approach for radio‑ablation after resection of breast ductal carcinoma in‑situ based on the BAT‑90
medical device. - A Novel Injectable Hydrogel Matrix Loaded with 90Y Microspheres for the treatment of Solid Tumours.
- In-vivo models for the performance and safety of BAT-90, a novel 90-Yttrium-based internal radiotherapy platform.
- Preclinical study of safety and efficacy in vivo of a yttrium-90 resin
microspheres glue formulation in a large animal model of pancreatic cancer. - Clinical Percutaneous image-guided radio-ablation using BAT-90 – initial results of ongoing first-in-man clinical trial.
Advantages of YntraDose®
For users
Nuclear physicians & interventional radiologists
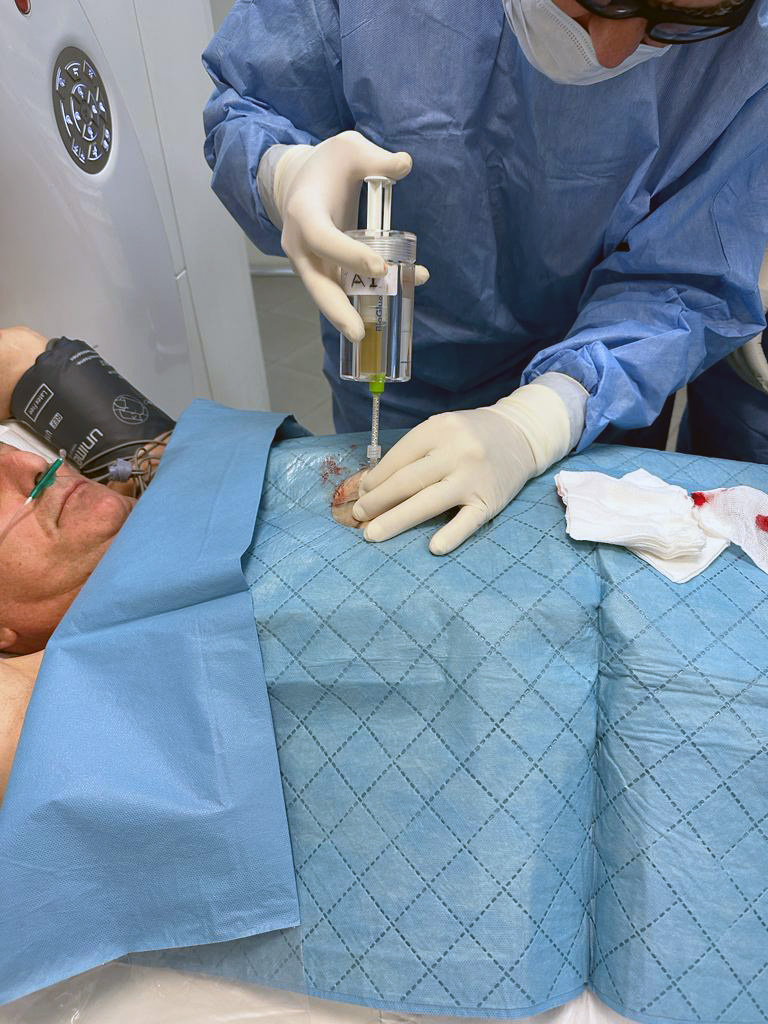
Easy to Use: YntraDose consists of a single percutaneous injection administered under CT guidance, delivering a fixed volume and dose to pancreatic lesions classified as unresectable locally advanced pancreatic ductal adenocarcinoma (uLA-PDAC). YntraDose is used as an adjunctive therapy in combination with standard of care (SOC) treatments such as FOLFIRINOX or gemcitabine/nab-paclitaxel
For patients
A solution exists
Safe: YntraDose is designed to avoid the need for general anaesthesia, to eliminate off-target radiation and with the ability to treat tumours near delicate organ structures
Convenient: Radiotherapy provided as a single day-case procedure as an outpatient
Minimally Invasive Procedure: Simple administration using an introducer/needle

For healthcare providers
Simple method
Accessible: Provides a radiotherapeutic treatment option in unresectable Locally Advanced PDAC where there is a huge unmet medical need
Cost Effective: No need for capital equipment and procedures are ‘non-DRG’ day-case
Protection
Intellectual
property
BetaGlue Medical possesses a wholly owned intellectual property estate across multiple jurisdictions. In addition to both granted and pending composition of matter, product by process and medical use patent claims, the company also owns a significant body of know-how. BetaGlue Medical, and YntraDose are registered trademarks of BetaGlue Technologies SpA.