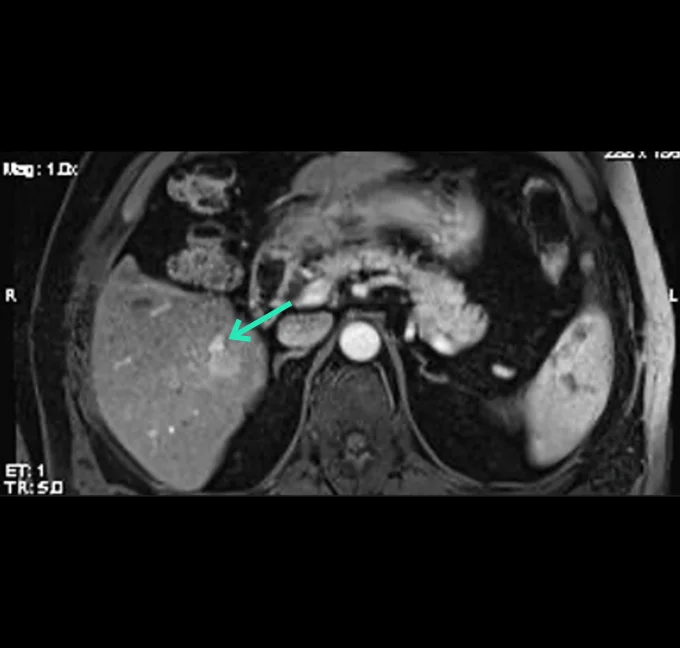
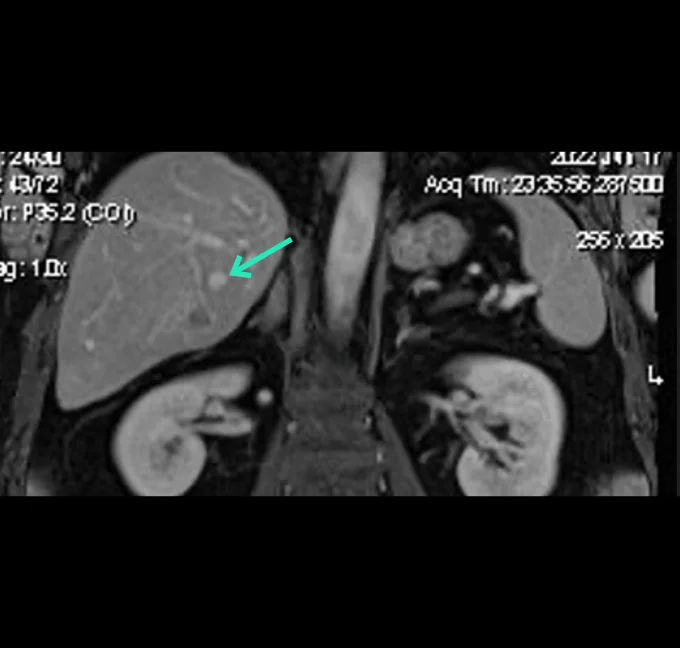
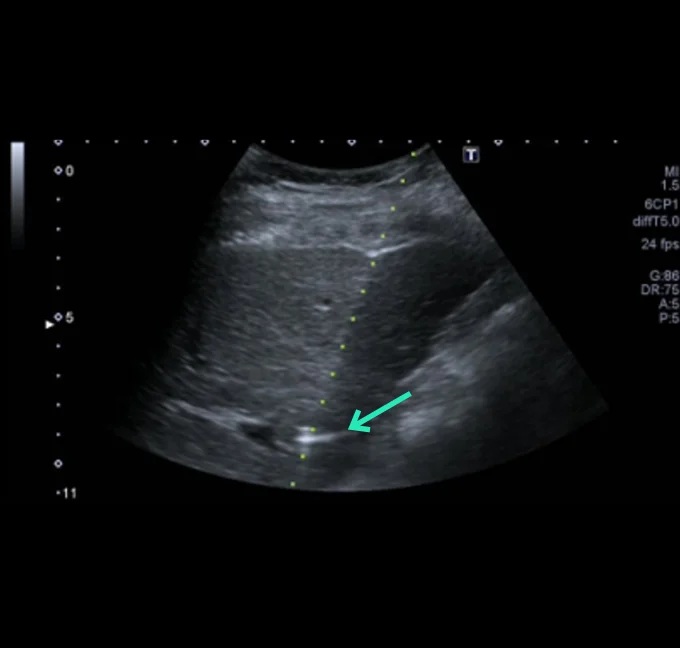
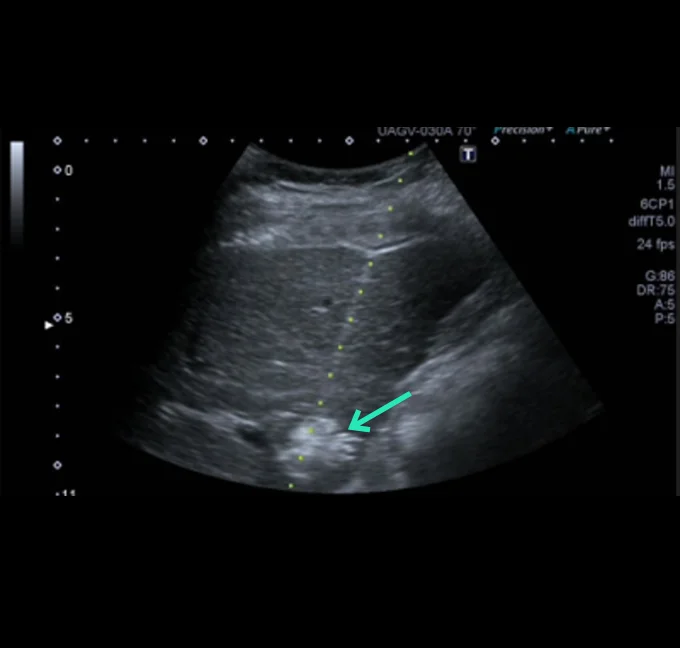
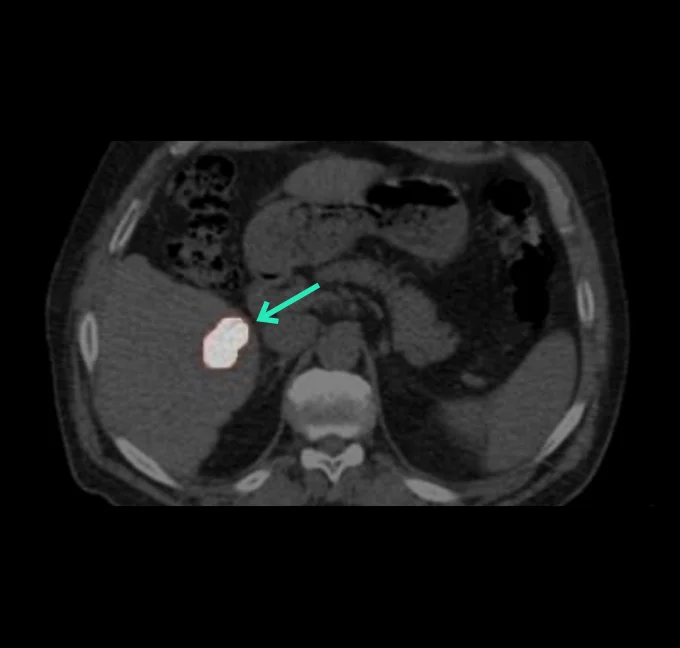
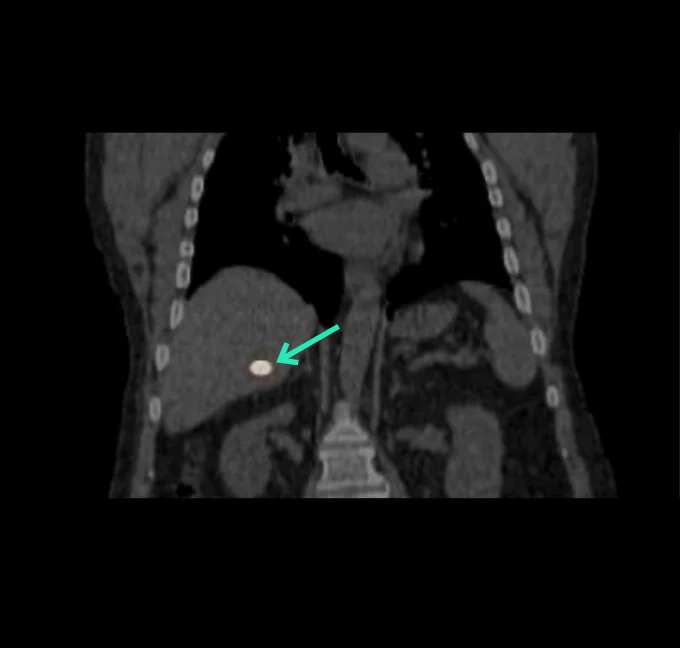
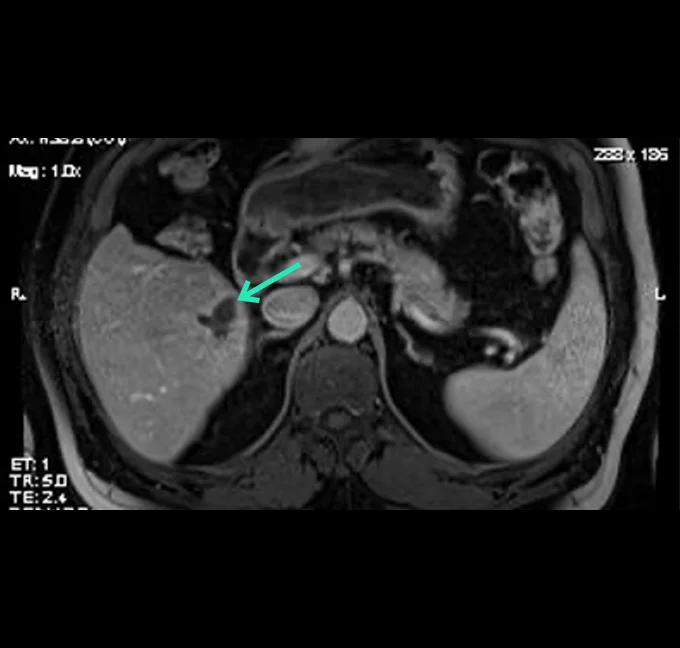
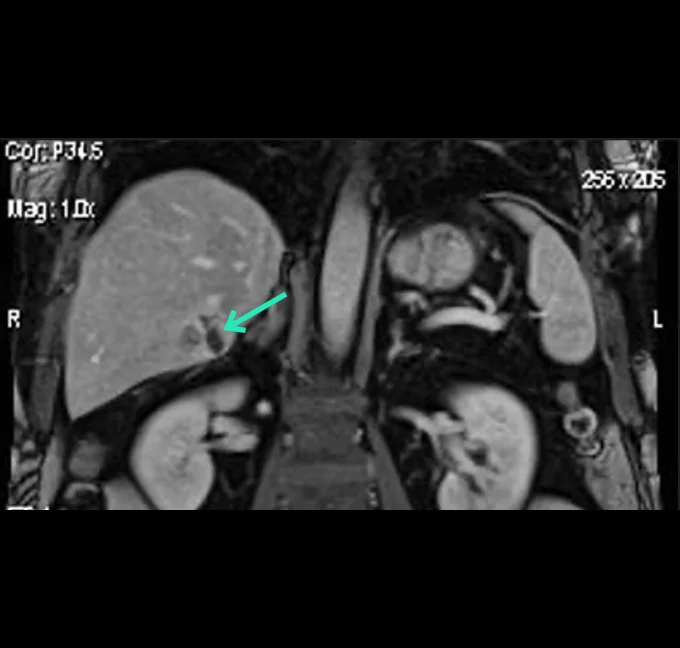
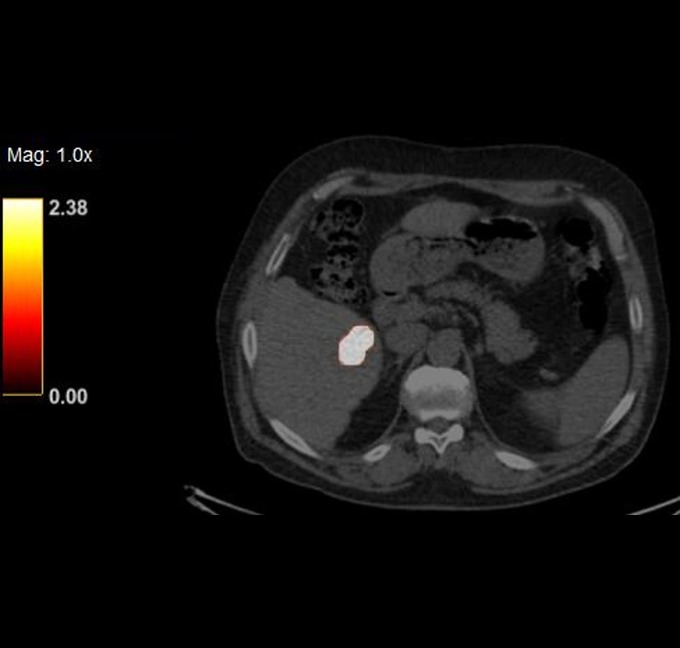
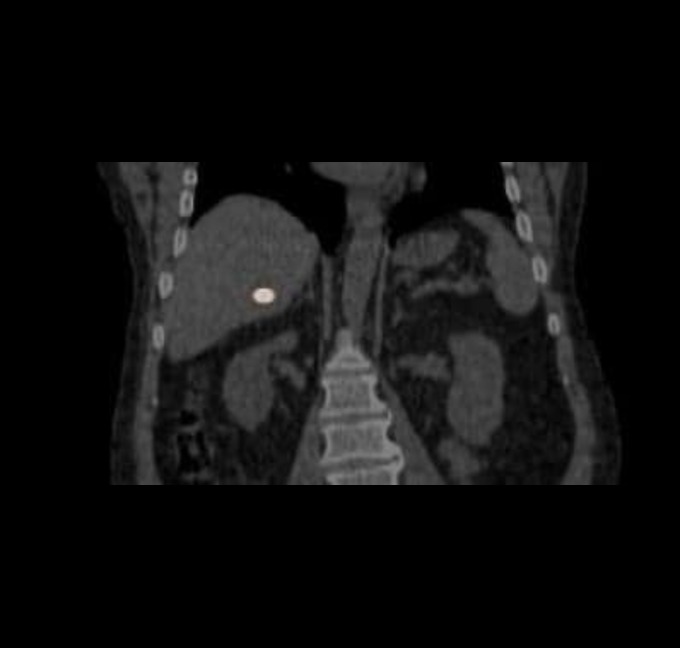
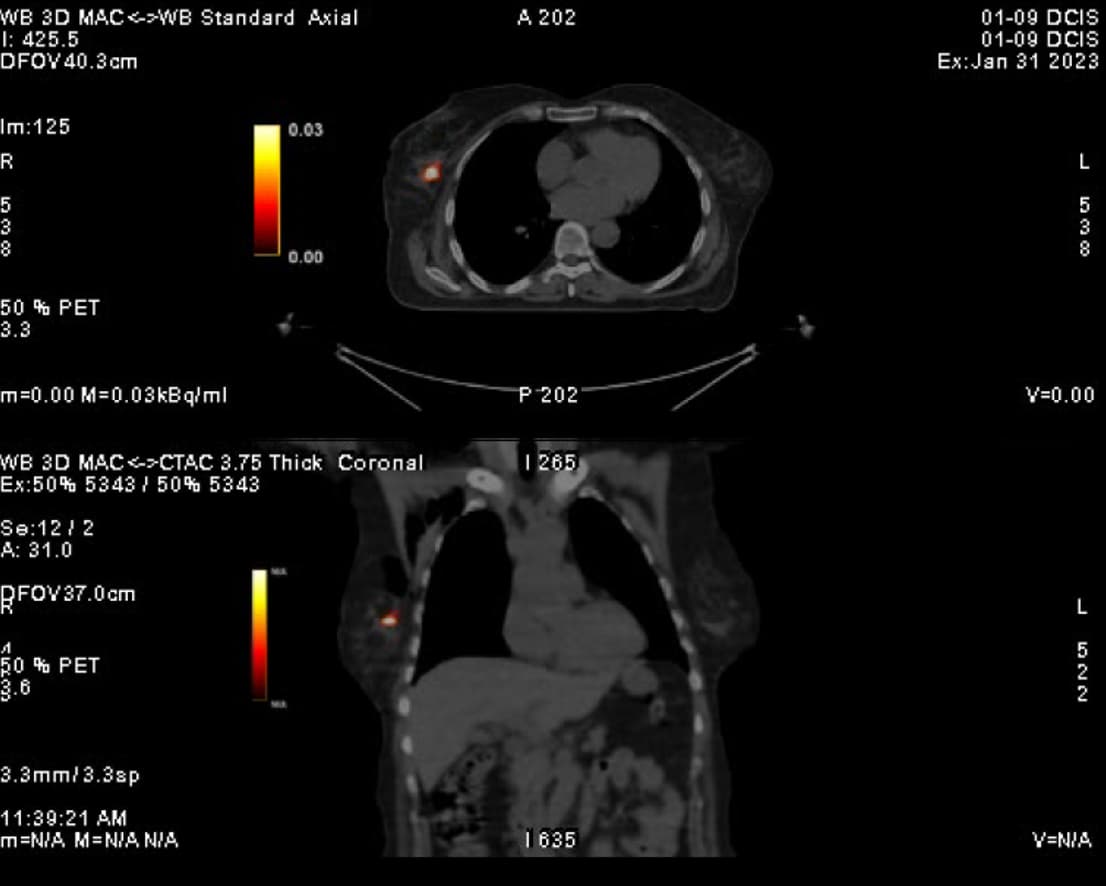
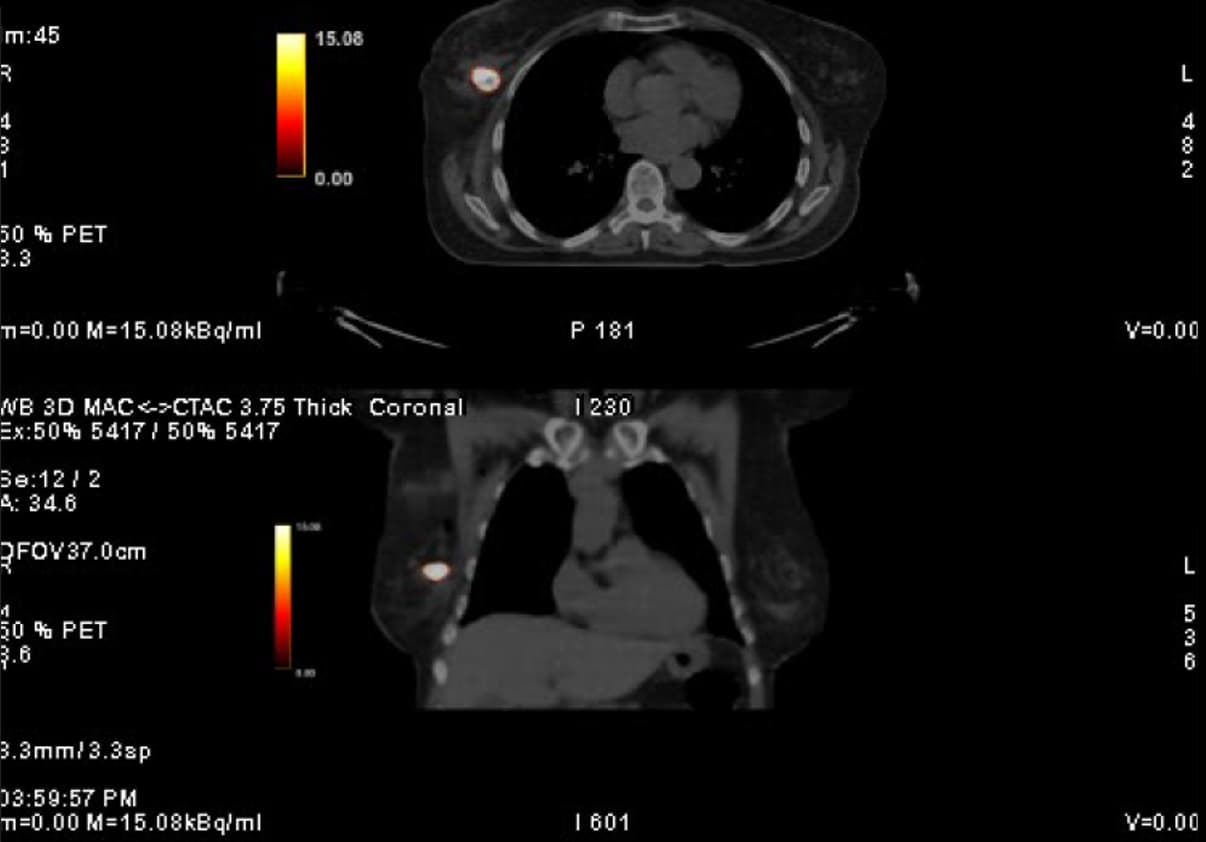
At the very beginning of the history of Interventional Oncology, ethanol injection, the only modality available at that time, was rather impressive, given that with simple percutaneous injections it allowed to achieve outcomes comparable to those of complex surgical procedures, albeit in selected cases.
The new technology of YntraDose, using Y-90, seems to be the natural evolution of the old ethanol injection, but applicable in a wide range of neoplastic diseases of different organs, with single applications and greater precision of guidance, given the great progress of imaging modalities. In summary, an actual ‘targeted therapy’ extensively feasible.
Prof. Luigi Solbiati